Accelerating towards a world without disease
or why we started Pheiron
500 years
That is, roughly, how long humanity will have to wait, at the current pace of drug development, for every known disease to have a cure. It is the ratio of uncured diseases to net new drugs our industry approves each year1. That’s twenty generations of patients waiting.
We started Pheiron because this timeline isn’t inevitable. We think it’s time to accelerate.
Making drugs is hard; Pioneering new biology is harder
Nine out of ten drug programs that reach human testing will fail. Six or seven of every ten drugs fail in Phase II. Everyone in the industry knows this.
Making drugs is hard because biology is hard. The human body is the most complex system we have ever tried to understand, and the honest answer to most of the questions is: we don’t know yet.
But biology risk is not evenly distributed. It concentrates on novelty.
Pioneering new biology, a new target, a new mechanism, a new disease hypothesis, multiplies biology risk and raises the odds of failure. The safe path is another me-too. The rational move in biopharma today is to stay close to what’s known.
But risk avoidance does not produce net new cures. Me-too’s do not help the patients who are still waiting.
Figure: At current pace, 20 generations of patients will have to wait for all diseases to be cured.
Every failed drug program had patients waiting for its cure
Investing in a novel biological hypothesis means betting on the validity of a long chain of experiments. Cell-based screening, animal experiments, safety / tox, even clinical trials, each is a probabilistic instrument trying to inform a single ground truth question: will this biology translate to humans?
Today, the industry has no systematic way of quantifying the probability that a drug program will succeed. We cannot quantitatively distinguish novelty likely to work from novelty likely to fail.
Our industry avoids novel biology because it lacks the capability to systematically underwrite biology risk.
Most conversations about fixing drug development focus on making experiments faster or cheaper. Better binders, higher-throughput screens, organoids.
But the economics of late-stage clinical trials are not going to change materially any time soon. A Phase III that enrolls thousands of patients, runs for years, and follows rigorous regulatory science will remain one of the most expensive things a human organization can attempt.
Capital for new drugs will remain finite. The lever that actually decides how much disease we cure, per dollar and per year, is not the cost of experiments. It is the selection of which programs to run and which biology to fund in the first place.
We as an industry need to stop funding failures and dramatically improve our ability to select winning biology.
Underwriting biology risk
Of course, every good scientist and every good portfolio manager is reaching for something like this, through their experience, their taste, their intuition. Nearly everyone in biopharma believes they have a better method for picking winners. But almost none of them benchmarks those claims, rationally and systematically against actual clinical outcomes.
Behind closed doors, in the portfolio strategy and IC meetings, decisions are taken based on “vibes and conviction”, trusting the experts, those “who know what’s good”. But just like algorithms replaced human economic intuition2, algorithms will replace human biological intuition.
Pheiron is built on one central belief: biology risk is quantifiable.
Certainly not deterministically, but far more rigorously than the industry does today. The substrate to do this exists. Human health data is exploding globally. Hundreds of experiments are published daily on top of decades of prior programs. What’s missing is a system that turns this data into systematic probabilities and updates them in real time as new evidence emerges.
Once we can quantify biology risk, we can underwrite it. We can distinguish novelty that is genuinely risky from novelty that only looks risky, and act on the difference. We can arbitrage mispriced opportunities, we can identify exploits. We can move with conviction on biology that the field has dismissed as too uncertain to fund.
The ability to anticipate what biology translates successfully to humans equals biological alpha.
At Pheiron, we are building AI to systematically underwrite biology risk, decision by decision, across experiments and inflection points. In other words, a way to engineer biological alpha.
How we engineer biological alpha
When assessing the probability of program success, it’s critical to distinguish biological risk from the corporate risk of execution. The cleanest trial in the world fails on a mechanism that doesn’t translate. The strongest biology will fail through an underpowered or badly designed trial.
Established methods exist to quantify execution risk by extrapolating from known biology: parametrized simulations to assess the trial design, its statistical power, the execution plan. The biology risk gets folded in implicitly, through the priors.
Most agentic research relies on what’s already known: synthesizing literature, integrating existing knowledge from the public domain. This works for grading established biology, such as me-too programs and late-stage trials with long paper trails, but cannot inform novelty (see our blog post on why LLMs are weak alpha generators3).
At Pheiron, we take a different approach: we build proprietary models and run primary analyses on multimodal human data (genetics, -omics, clinical records, observational and interventional cohorts, spanning millions of lives) to generate novel and comprehensive human evidence. The question we answer, on every program: is this mechanism causal to the disease endpoint?
Our platform treats biology risk the way an underwriter treats any other risk: by quantifying and measuring meticulously against what matters: clinical outcomes.
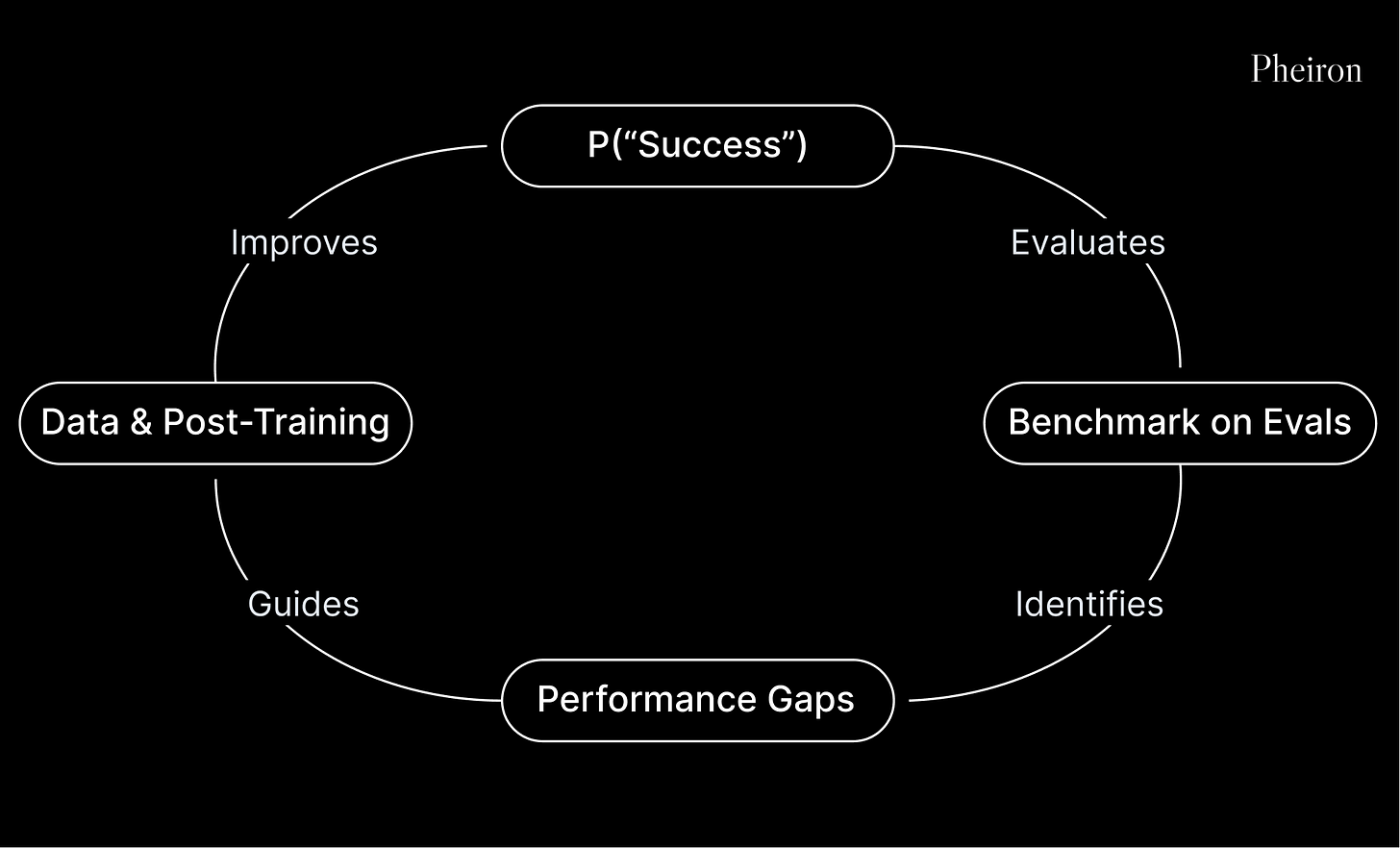
We construct retrospective and prospective evals to hillclimb biological alpha. We backtest on historical drug approvals and forward-evaluate on prospective approvals; we quantify the alpha that a given method, data source, or model adds to our calls, and iterate.
In backtests, our proprietary models and primary analyses capturing biological risk, enrich for successful programs at roughly ~20x over the base rate4.
Figure: Feedback loop for hillclimbing biological alpha.
We invite you to join us
So far, we’ve been building with partners who put the platform to work. Some have started programs on the back of what we surfaced, others have killed programs. But we’re just at the beginning.
Today, we’re entering the next phase. We are publishing our first set of public predictions on upcoming clinical trial readouts. Each carries two measures: Biological Support, our score for the underlying biology, and probabilities of trial success across the trial’s endpoints. Together, they let you see which lever is doing the work. You can read them at pheiron.com/predictions.
We are doing this for three reasons: to learn faster, to hold ourselves publicly accountable for the claims we make, and to invite the rest of the industry to do the same.
Accelerating towards a world without disease
If we are right, three things follow. They are the things we care about most.
First, pursuing novel biology becomes an economic necessity. When the industry can distinguish high-risk novelty from low-risk novelty and pursue the latter deliberately, the opportunity surface bends. That is how real progress happens.
Second, smaller opportunities become fundable. When risk can be priced, the risk-reward calculus no longer demands a blockbuster for a drug to be worth pursuing. Subtypes become viable. Rare diseases become viable. Personalized medicine is within reach.
Third, and this is the only measure that ultimately matters, we reach cures faster. It will still take time. But the 500-year timeline compresses. Every program not funded that was set up to fail is the opportunity to fund a program that might succeed. Every year we shave off that horizon is decades of suffering we did not let happen.
We believe that the missing piece is a disciplined, quantitative way to underwrite biology; this is the single most important lever towards a world without disease.
Pheiron is an applied AI lab dedicated to biological alpha. We use it to select and develop our own drug programs: identify mispriced opportunities, execute with conviction, and compound that advantage program after program to dramatically increase the drug-per-dollar output. This will allow us to break Eroom’s law5 and accelerate towards a world without disease.
If you’re a patient, or love someone who is, the wait you’re living through isn’t inevitable.
If you are a scientist, a drug hunter, a decision maker, a capital allocator, or someone who has spent long nights wondering if biology will translate: we would like to work with you.
Five hundred years is too long. Let’s get to work.
References
There are ~10,000 known diseases. 40-60 new drugs are approved per year, but of those only 10-20 are genuinely new, the rest are me-too’s aimed at market share not novelty.
Algorithmic trading, the replacement of human economic intuition with computers, gave rise to Renaissance Technologies, which is widely regarded as the most successful fund and arguably one of the most valuable technology platforms of all time.
Read our blog post on the ability of LLMs to predict unseen trial outcomes. Naive LLMs are weak alpha generators. https://blog.pheiron.com/p/llm-bench
Retrospective analysis on phase transitions of target-indication rates. Technical Report in preparation. For comparison: human genetic evidence is known to increase a program’s probability of success by 2 to 4x (Minikel, 2024)
Eroom’s Law is the observation that drug discovery is becoming slower and more expensive over time, with the inflation-adjusted cost of developing a new drug roughly doubling every nine years since the 1950s.